Business Development
We finalised the license agreement with the University of Zurich. This license gives us the exclusive right to use on the patent application necessary to commercialize the Tuberculini product. We have made progress with potential collaboration partners in India, who are interested in our technology.
Company Development
In our last General Assembly we have created the possibility to conduct future General Assemblies in digital format in line with the move to digitize business processes to increase efficiency.
Product Development
Software Development
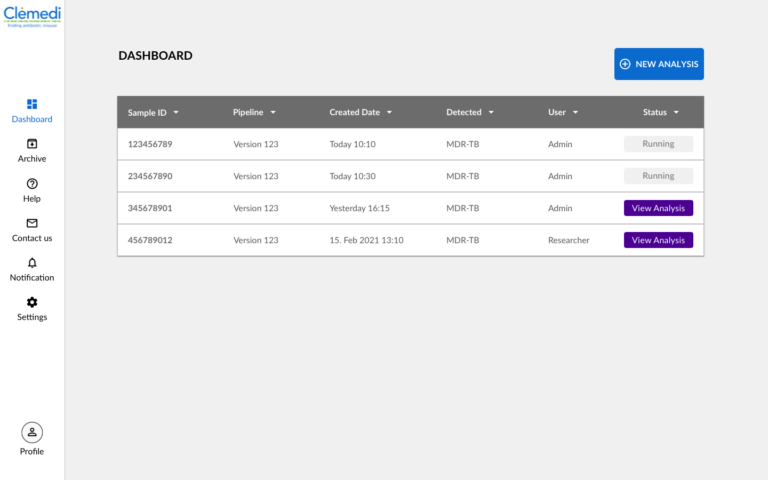
We have translated the user interface templates that were validated in a small-scale user study into an interactive and clickable online application that can be hosted in the cloud.
Below you can see mockups of the registration screen as well as the dashboard that shows a list of analyses and the menu for the functionality of the application.

Lab Development
Work packages financed by the Innosuisse Grant we obtained last year are on track. Experiments are ongoing at our partner site at the University of Bern. We also have contracted other international sites in Italy, Pakistan and Uganda to support in validating our Tuberculini product.
Fundraising
We were able to close a convertible round of CHF 385k from several Angel Investors end of June. The financing, will enable us to reach our the design freeze of Tuberculini, the first application on our Disease Management Platform as well as plan the upcoming clincial study.

